
Back on January 4, 2018, the industry was in a slight tailspin due to then acting Attorney General Jeff Session’s (renowned marijuana hater) rescinding of all marijuana enforcement guidance from the Department of Justice (“DOJ”). Reactions in the media ranged from treating the Sessions announcement as nothing more than an attempt to frighten the cannabis industry to claiming that it was the first step in an organized crackdown of the marijuana industry that could affect cannabis businesses and users. Both possibilities are arguably realistic. And the drama that followed Sessions’ moves was pretty satisfying, including when Cory Gardner vowed to (and did) block DOJ appointments until the issue was resolved in favor of the states, culminating in a deal with President Trump to back off of state-legal marijuana. However, now that Sessions is out at the helm of the DOJ, industry folks can breathe a little easier where new Attorney General nominee William Barr has gone on record stating that state-law abiding cannabis businesses will not be prosecuted by the DOJ and essentially that the 2013 Cole Memo will be back from the dead. In rescinding all DOJ guidance on marijuana enforcement, Sessions torpedoed the famous 2013 Cole Memo, which outlined eight specific enforcement priorities of the DOJ in states with legal marijuana and which, between the lines, indicated that “robust” state regulations would keep the DOJ at bay regarding enforcement of the federal Controlled Substances Act. After that memo, entire states built their comprehensive cannabis licensing and taxation systems on those eight enforcement priorities, ensuring that compliance restrictions and barriers to entry were strong enough to support the same. Instead, Sessions put in place the “Sessions Memo,” which was short on specifics. It doesn’t contain an outright directive ordering U.S. Attorneys to go after marijuana businesses. It simply withdraws all of the earlier marijuana-specific guidance memoranda and directed U.S. attorneys to treat marijuana sales like any other federal crime. The withdrawn memos include, the 2013 Cole Memo, the February 2014 Cole Memo that extended low enforcement priority status to apply to banking activities (although the FinCEN guidelines are, importantly, still alive); and the 2014 Wilkinson Memo that was a sort of Cole Memo for tribal lands. Right now, U.S. attorneys have full discretion to determine to what extent they can/should enforce federal law in the context of marijuana crimes in states with legalization and medicalization–which they always had anyway–but the 2013 Cole Memo helped them prioritize certain marijuana issues across the DOJ. In his memo, Sessions referred to the principles of enforcement in the U.S. Attorneys’ Manual, but that document reinforces the level of discretion and authority that each U.S. attorney has already. The Cole Memo was ultimately useful in providing a consistent nationwide federal policy. Under Sessions Memo, we are back to the days of having potentially 93 different enforcement policies — one for each U.S. Attorney. To date, there haven’t been any reported incidents of the Feds going after state-law compliant cannabis operators in states that have legalized and regulated. A new sheriff is coming to town though, and that could be a very good thing for the momentum of state-by-state legalization in that states will better know what to expect from Big Brother as will marijuana businesses and their investors. William Barr may end up becoming a very unlikely helper when it comes to state-legal cannabis. He was Bush I’s attorney general from 1991-1993, and he’s a dyed in the wool conservative who, as Attorney General, was “tough on crime” and put many, many people in prison. As reported by Marijuana Moment, Barr in a mid-January hearing with Congress testified that:
While Barr also testified that he wouldn’t go “after companies that have relied on [2013] Cole memorandum . . . ,” he also didn’t completely kowtow to state legal cannabis. He further testified that “we either should have a federal law that prohibits marijuana everywhere, which I would support myself because I think it’s a mistake to back off marijuana. However, if we want a federal approach—if we want states to have their own laws—then let’s get there and get there in the right way.” In reading the tea leaves, it sounds like, personally, Barr would have no issue with continuing the War on Drugs as it relates to cannabis. As a department under his watch and command, however, the DOJ probably wouldn’t spend time and valuable resources on state-legal operators — even if Barr is concerned that the current dynamic is breeding “disrespect for the federal law.” Reasonable minds can differ, but I’d say that most cannabis operators and states are very mindful of federal law enforcement and it’s really Congress, the DOJ, and the President to blame for creating legal confusion because of varied enforcement over the years. In the end, Barr’s testimony ultimately serves to show the country that Congress has been woefully impotent and ignorant when it comes to cannabis as a whole and especially as the topic relates to states’ rights. What’s good to know though is that if Barr is confirmed, we’re very likely returning to the 2013 Cole Memo principles, which will at least create a political atmosphere of certainty in that the DOJ has bigger fish to fry than state-legal marijuana. Right now, Barr is pretty much a lock for U.S. Attorney General, so hopefully he’ll make good on his cannabis compromises. Source: https://www.cannalawblog.com/icymi-u-s-attorney-general-nominee-likely-wont-harsh-your-mellow/ The following blog post ICYMI: U.S. Attorney General Nominee (Likely) Won’t Harsh Your Mellow was initially seen on FeliciaSullivan.com from https://www.feliciasullivan.com/2019/02/12/icymi-u-s-attorney-general-nominee-likely-wont-harsh-your-mellow/
0 Comments

This post is part one of two on how the State of New York is regulating CBD. Last week, New York City’s Department of Health (“DOH”) quarantined a number of edible products that contained hemp-derived CBD (“Hemp-CBD”) and announced that Hemp-CBD would not be allowed in food products in the City. Eater first broke the story, but the crackdown made national news with the Wall Street Journal, the New York Times, NBC, and Fox all publishing stories on the event. It is unsurprising that the DOH action drew such coverage. CBD is massively popular, New York City is the largest city in the United States, and the story is compelling because the DOH actually sent out agents to quarantine products, rather than simply issuing a statement. It seems to be this last point that garnered national attention based on the relative lack of coverage a very similar story garnered back in December 2018. On December 18, 2018, shortly before the signing of the 2018 Farm Bill, the New York State Department of Agriculture and Markets (the “Department”) issued a series of frequently asked questions (“FAQs“) and a CBD Processor Template Agreement (“CBD Agreement“) that were both focused on Hemp-CBD. Unlike the DOH, the Department did not take any enforcement action. Coverage of the FAQs and the CBD Agreement was sparse. Now that New York City is taking action, it’s time to dig into the state’s position on Hemp-CBD. The Department oversees New York’s industrial hemp program, which was promulgated under the 2014 Farm Bill. Rather than issuing licenses or permits, the Department enters into research agreements with individuals and companies who wish to process hemp into commercial products. The Department uses a number of template agreements available online. The CBD Agreement applies to processors who wish to create Hemp-CBD products intended for human consumption. The FAQs and the CBD Agreement make it clear that the Department is intending to treat Hemp-CBD as a dietary supplement. The FAQs state that an individual cannot “sell any item for human consumption that has CBD as an ingredient unless” the two following standards are met:
The FAQs elaborate on its dietary supplement standard:
This dietary supplement classification applies to any “product that is a combination of ready-to-eat food with additional CBD infusions or CBD extracts, such as CBD chocolate syrup or CBD soda or CBD-infused frosting drizzled cookies.” The FAQs also make clear that they apply to products from other states: “products made from industrial hemp that are sold in NYS must meet NYS standards, regardless of where the product is processed or manufactured.” It should be noted that the Department acknowledges that its jurisdiction over Hemp-CBD is limited. For example, the Department does not require businesses to apply to the Department to add pre-manufactured Hemp-CBD to another product such as a topical or to develop products using CBD is sourced from another state. There is also no requirement to obtain any authorization from the Department to sell Hemp-CBD products. However, if the product is sold anywhere in New York State, it must comply with dietary supplement standards. You can agree or disagree with the New York City DOH’s decision to start quarantining CBD in food, however, it does seem to be inline with the Department’s guidance. The Department is a state agency and the DOH is a city agency so DOH’s decision may be it showing deference to the Department. Later this week, I’ll analyze the CBD Agreement and how it provides additional insight into the state’s position on Hemp-CBD. Source: https://www.cannalawblog.com/beyond-the-crackdown-new-york-classifies-hemp-cbd-as-a-dietary-supplement-part-one/ The following post Beyond the Crackdown: New York Classifies Hemp-CBD as a Dietary Supplement (Part One) Read more on: FeliciaSullivan.com from https://www.feliciasullivan.com/2019/02/11/beyond-the-crackdown-new-york-classifies-hemp-cbd-as-a-dietary-supplement-part-one/
On February 1, it was reported that the World Health Organization (WHO) made some significant and long overdue recommendations with respect to cannabis. Those recommendations have not been formally released, but we expect that to happen soon. If adopted wholesale by the United Nations (UN), the recommendations will have a significant impact globally as to controls placed on cannabis and its constituent parts. It is important to note that the WHO is not recommending the wholesale legalization of marijuana. Therefore, no one should expect the doors to swing wide on international cannabis trade overnight. Still, the WHO development is welcome news after nearly 60 years of unmerited and unexamined prohibition of cannabis under international law. The WHO recommendations are reported as follows:
So what would all of this mean? First, cannabis containing more than trace amounts of THC would still be controlled. Whole plant marijuana would no longer be in the same class of drugs as heroin and fentanyl, but it would not be eligible for trade in the same way as coffee or even tobacco. Second, concentrated preparations of THC would be controlled more strictly than flower, but not at draconian levels. Third, penalties for distribution and possession of cannabis in any form would significantly decrease. And fourth, CBD would be treated like bonbons. In all, the WHO approach is measured and scientific, seeking to isolate various parts and preparations of the plant, and distinguish their effects. When it comes to implications for U.S. law, the WHO’s assessment of CBD could have the most immediate impact. Readers of this blog may recall that the U.S. Drug Enforcement Administration (DEA) has taken the position that the U.S. would “not be able to keep obligations under the [Single Convention] if CBD were decontrolled under the CSA”. The Food and Drug Administration (FDA) ultimately fell in line with DEA’s interpretation, scheduled Epidiolex (an approved CBD drug), and recently issued a public statement warning that it is unlawful “to introduce food containing added CBD … into interstate commerce.” If the WHO recommendation is adopted by the UN, though, the FDA may reverse course quickly. When FDA agreed to schedule Epidiolex, it advised: “If treaty obligations do not require control of CBD, or the international controls on CBD … are removed at some future time, the recommendation for Schedule V under the CSA would need to be revisited promptly.” Presumably, state health authorities would fall in line with the FDA’s ruling, and we would stop seeing things like last week’s raids in New York and Maine. At some point this year, it is likely that the UN will vote on the WHO’s cannabis rescheduling recommendations. A really, really, really interesting question is where the United States will cast its lot on that momentous day. The U.S. has always been an international hardliner when it comes to cannabis, even as a vast majority of its states have legalized marijuana for medical or recreational use. These days, though, things are changing fast – both domestically and worldwide. For more on cannabis and international law, check out the following:
Source: https://www.cannalawblog.com/the-u-n-inches-closer-to-cannabis-reform/ The article The World Health Organization Steps Up on Cannabis was first published to Felicia's Smokin' Blog from https://www.feliciasullivan.com/2019/02/10/the-world-health-organization-steps-up-on-cannabis/ Find your perfect wake and bake strain and even the dreariest Monday morning can become an absolute joy. Well, maybe not a joy as such, but at least a little less painful. Which begs the question – what makes the ideal strain for a perfect wake and bake? The answer – the combination of fabulous […] The post The Best Wake and Bake Strains Money Can Buy appeared first on Legalize it. We Think So. Source: https://www.liwts.org/cannabis/the-best-wake-and-bake-strains-money-can-buy/ The Best Wake and Bake Strains Money Can Buy is available on https://www.feliciasullivan.com/ from https://www.feliciasullivan.com/2019/02/10/the-best-wake-and-bake-strains-money-can-buy/ 
State trademarks for cannabis goods and services have been an ongoing saga in California that we have written about extensively. For a little background, until January 1, 2018, obtaining state trademark protection in California was not possible due to Sections 14270-14272 of the Model State Trademark Law of the California Business and Professions Code, which are simply titled “Miscellaneous.” Section 14272 states the following:
However, in December of 2017, the California Secretary of State’s Office announced that customers would be able to register cannabis-related trademarks or service marks so long as the following requirements are met:
The Secretary of State’s Office has reiterated that it will only accept applications insofar as the goods and/or services in question fit within an existing classification code from the USPTO’s Identification of Goods and Services Manual. Therefore, it is easy to register for things that fit squarely within the USPTO specifications, like retail services. Cannabis goods are a bit more problematic, although we have developed strategies to protect these as well. All of this has been based on administrative policy declared by the Secretary of State’s Office, not on legislation or a change to the California Business and Professions Code, but Senate Bill 185, which was introduced on January 30, 2019 and just went to committee, would change that. SB 185 notes that existing law in California provides for registration of trademarks where the classification of goods and services for those marks conforms to the classifications adopted by the USPTO, but proposes that for marks for which a certificate of registration is issued on or after January 1, 2020, applicants would be authorized to use “specified classifications for marks related to cannabis, including medicinal cannabis, goods and services that are lawfully in commerce under state law in the State of California.” Designated classifications of goods for cannabis products would streamline the process for trademark registration in California and provide cannabis companies with greater security regarding the enforceability of their registrations. Additionally, SB 185 provides that the Department of Food and Agriculture, in conjunction with the State Department of Public Health and pursuant to MAUCRSA, must establish a certification program for cannabis and manufactured cannabis products comparable to the federal National Organic Program and the California Organic Food and Farming Act. As we’ve written before, it is not permissible to use an organic designation on cannabis products unless that designation is pursuant to state law or pursuant to a private certification, since the U.S. Department of Agriculture generally regulates that certification under the Organic Foods Production Act. SB 185 lays the groundwork for some important improvements to the way cannabis companies protect their brands and the establishment of an organic certification program will benefit both companies and consumers. We’ll be following this bill closely and hope it doesn’t stall in committee. Stay tuned! Source: https://www.cannalawblog.com/california-cannabis-trademarks-could-get-a-legislative-boost/ The blog article California Cannabis Trademarks Could Get a Legislative Boost Find more on: The Felicia Sullivan Blog from https://www.feliciasullivan.com/2019/02/09/california-cannabis-trademarks-could-get-a-legislative-boost/ Realistically, it’s impossible to avoid building a somewhat developed and strong tolerance to THC over time. It’s the same with any drug – the more you use it, the greater the resistance your body develops to it and the more you need. Some stay in lightweight territory for their entire lives, but most will inevitably […] The post 8 Ways to Deal With an Immense THC Tolerance appeared first on Legalize it. We Think So. Source: https://www.liwts.org/blog/8-ways-to-deal-with-an-immense-thc-tolerance/ 8 Ways to Deal With an Immense THC Tolerance was initially published on Felicia Sullivan from https://www.feliciasullivan.com/2019/02/09/8-ways-to-deal-with-an-immense-thc-tolerance/
Earlier this year, the Food and Drug Administration (“FDA”) began seizing various cannabidiol (“CBD”) products from store shelves. These enforcement actions reflected the implementation of the agency’s position that CBD, regardless of the source from which it is derived, cannot be lawfully sold for human consumption. A few states, including states that have adopted industrial hemp pilot research programs under the 2014 Farm Bill, now seem to have embraced this FDA position by banning certain CBD-infused products from local stores. Last Friday, several New York restaurants, bakeries, and bars were forced to stop selling CBD-infused foods and drinks. Officials with the state Department of Health confiscated those products, marking them as “embargoed.” The embargo process consists of identifying, itemizing and removing products. The Department has yet to issue a public statement or to provide further information on these actions, but it appears that state health inspectors explained to the affected business owners that CBD could not be used as a “food additive”. This argument was similar to that used by the Maine Department of Health and Human Services (“DHHS”). Earlier last week, Maine health authorities began notifying businesses that they were required to remove all CBD-infused foods, tinctures, and capsules from their shelves. Relying on an internal report by the state Attorney General’s Office, which concluded that CBD could not be used in mass-market food until Maine’s hemp pilot program receives federal approval pursuant to the 2018 Farm Bill, the Maine DHHS determined that CBD was an unapproved food additive that the FDA does not recognize as safe. Section 201(s) of the Food, Drug, and Cosmetic Act (“FD&C Act”) defines “food additive” to encompass any substance that may reasonably be expected to directly or indirectly affect or become part of a food. Until a food additive is tested and reveals to be safe for its intended use, it is deemed unsafe. A food additive is considered safe if there is a reasonable certainty in the minds of competent scientists that a substance is not harmful under its intended use and condition. If a food additive is added to a food prior to FDA approval, its presence renders the food adulterated and subject to enforcement action. Because the FDA has yet to approve CBD as a food additive, is it accurate that Hemp-CBD products, particularly edibles and infused drinks, would be deemed unsafe under the FD&C Act. Although the state health authorities embargoed CBD edibles and other CBD products used for human consumption, they told affected business owners that they could continue selling CBD products that “could be smoked, vaped, worn as a patch or applied as lotion.” This is because cosmetics and smokable products are subject to less onerous FDA regulations than foods and dietary supplements. Nevertheless, this is all likely to change because the FDA will undoubtedly release a plan to regulate Hemp-CBD products soon. These recent enforcement actions in places like New York and Maine threaten to spook this fast-growing industry and should remind industry players that business and legal considerations surrounding the sale of Hemp-CBD products are in a constant state of flux. We will continue to monitor this issue and update you on other states CBD enforcement actions. Until then, don’t hesitate to contact our offices for additional information on the legal status of state and federal Hemp-CBD law and policy. Source: https://www.cannalawblog.com/state-crackdowns-against-cbd-products-have-begun/ The post State Crackdowns Against CBD Products Have Begun is republished from https://www.feliciasullivan.com/ from https://www.feliciasullivan.com/2019/02/08/state-crackdowns-against-cbd-products-have-begun/ As you may have noticed, recently we’ve added a wide range of magic mushrooms from Freshmushrooms to our online catalogue. While growing them at home is easy, it does require carefully following a few important steps to ensure everything works as it should and you can enjoy a successful, problem-free harvest of your own magic mushrooms. In this article we present a concise guide to using our mushroom growing kits in a simple and effective way. You’ll be surprised at how easy it can be! 
A small forest of Amazonica mushrooms
Cultivating Freshmushrooms magic mushrooms step by stepGrowing your own mushrooms like the famous Psilocybe Cubensis Panamericana, the classic Golden Teacher or the legendary Psicolybe Mckennaii is now easily within reach, and as you’ll see below, the team at Freshmushrooms couldn’t have made it easier thanks to this handy guide! These mushroom cultivation kits include everything you’ll need to get excellent crops in just a few weeks, without complications or risks, and with shrooms loaded with psilocybin. Let’s do it! Step 1:Open the box and remove the contents: the mushroom growing kit, the plastic bag and the paperclip. Remove the lid of the kit’s plastic container, rinse it well with clean water and store it properly, you’ll need it later (you can keep it safe inside a clean zip-close plastic bag, for example). Step 2:Place the plastic container containing the substrate (without the lid) into the included plastic bag. Slip the opening of the bag underneath the container, with the micro-perforations of the plastic bag facing up. This ensures the kit has sufficient air circulation (the opening of the bag is largely closed now, leaving the plastic container “wrapped” in the breathable bag). 
The plastic container must be put in the bag Step 3:As soon as you see signs the first mushrooms appear in the substrate, reposition the bag vertically, upright, allowing the kit enough space for the mushrooms to develop. Notice that we still haven’t used any water in any of the steps! If everything works correctly, the first mushrooms should be formed in a week. Step 4:Put the bag with the container in a place with enough daylight (but never in direct sunlight!) and at a temperature between 18 and 23ºC. Mushrooms grow at temperatures above 15ºC, although 23ºC is perfect for larger harvests. If necessary, accessories such as the RootIt heating mat are ideal to maintain an ideal and constant temperature for the mushrooms, so it’s not necessary to heat the entire room where you’re growing them. Some people grow them close to a window in summer and put them inside a small heated greenhouse during winter with some 24W T5 Fluorescent tubes. If you want to keep everything private, put everything inside a small grow tent for clones. Step 5:As soon as you see the first fully formed mushrooms, you can open the bag slightly to avoid a build up of condensation. At this point, excess moisture can be detrimental to the development of mushrooms. The first mushrooms should be ready to harvest about a week after having seen the appearance of the first “buttons” in the substrate (step 3). Remember that you can monitor temperature and humidity at all times with the help of a digital thermo-hygrometer. 
After just a few days we can see the first mushrooms Step 6:The moment to harvest the mushrooms is very important. You should examine the mushroom’s cap; If it’s swollen but the underside has not yet opened to allow you see the gills that contain the spores, then it’s the perfect moment. It’s important to harvest them before they open, leaving the gills exposed, thus releasing the spores. Step 7:Now, some excellent news for you… you can reuse the mushroom kit without needing to inoculate with fresh spores! It’s that easy! Simply harvest all the mushrooms and fill the container with clean water, a process called cold shock that promotes the formation of new life in the mycelium and ensures te substrate has enough water to develop a new crop of mushrooms. 
We’re soon harvesting the first mushrooms Step 8:Cover the container with the lid you stored in step 1 and let it rest for 12 hours. Once this time has elapsed, open a corner of the lid and pour out the excess water not absorbed by the substrate. Now it’s ready to go again, you just have to follow the instructions again from step 1. You can reuse the kit several times, as long as you are scrupulously clean and careful after each step. Tricks for mushroom cultivation
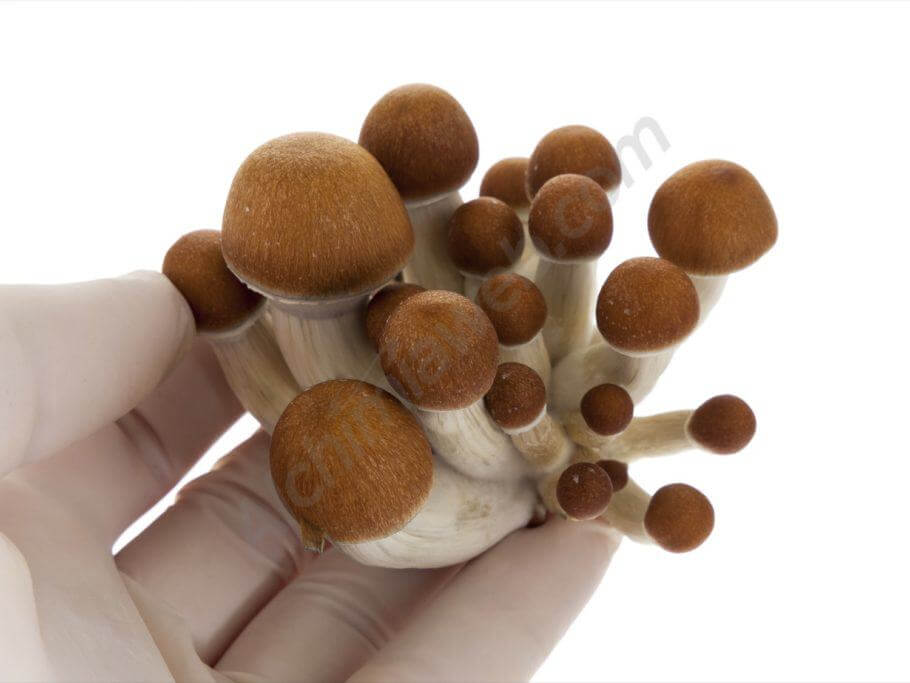
Cluster of Colombian Psilocybes
Hopefully we’ve shown clearly how to grow these mushrooms; As you can see it’s very simple and you can also reuse the mushroom kits several times, getting several harvests out of each kit. Don’t hesitate to leave us your comments or questions below, we’ll happily respond. Happy harvests! The post How to grow magic mushrooms from Freshmushrooms appeared first on Alchimia blog. Source: https://www.alchimiaweb.com/blogen/how-grow-magic-mushrooms/ The post How to grow magic mushrooms from Freshmushrooms was first published to Felicia Sullivan's Blog from https://www.feliciasullivan.com/2019/02/08/how-to-grow-magic-mushrooms-from-freshmushrooms/ TV host and longtime medical cannabis patient and advocate Montel Williams joins the show to discuss how cannabis has helped his health, and why he's started his own line of products in partnership with Cura Cannabis. The post What Are You Smoking Episode 69: PBud and Chemdog appeared first on Leafly. Source: https://www.leafly.com/news/podcasts/what-are-you-smoking-69-pbud-chemdog The article What Are You Smoking Episode 69: PBud and Chemdog was initially seen on www.feliciasullivan.com from https://www.feliciasullivan.com/2019/02/07/what-are-you-smoking-episode-69-pbud-and-chemdog/
The bill requires Washington comply with the 2018 Farm Bill, which removed industrial hemp from the federal Controlled Substances Act, and which provides for state- and tribe-level programs for the cultivation of industrial hemp. As such, the Washington State Department of Agriculture (“WSDA”) would need to submit a plan to US Department of Agriculture (“USDA”) pursuant to the 2018 Farm Bill. The plan would need to cover licensing, THC testing, enforcement, and a host of other topics required under federal law. It’s too early to tell whether SB 5719 will ultimately become law, but it’s worth keeping an eye on at this point (and will be covered in our upcoming free CBD Webinar on February 21 at 12:00 PST). Food and Hemp SB 5719 would repeal Washington’s Industrial Hemp Research Program (RCW 15.120 et seq.) completely. This program was created under the 2014 Farm Bill, which was fairly light on details and limited to hemp cultivation for “research” purposes. Washington’s hemp program is fairly limited and has never really taken off like it has in Colorado, Kentucky, and Oregon. Washington’s hemp law is also extremely confusing thanks to RCW 15.120.020 which prohibits the “production of any part of industrial hemp, except seed, as food, extract, oil, cake, concentrate, resin, or other preparation for topical use, oral consumption, or inhalation by humans[.]” This provision only refers to the production of hemp, not the sale of hemp products, including widely popular hemp-derived CBD (“Hemp-CBD”) products. However, it has also stunted the retail sale of hemp products intended for human consumption under the idea that if it’s illegal to produce hemp for human consumption it is also legal to sell hemp products for human consumption. This would no longer be an issue if SB 5719 passes. However, SB 5719 does come with some ambiguity. Under, SB 5719 “CBD and CBD products derived from hemp are considered a food product that must be tested and treated in accordance with other agricultural crop derived food products for human and animal consumption.” This could lead to confusion. For example, a Hemp-CBD topical cream would still have to be “tested and treated” as food. That is inconsistent with FDA regulation which treats food and cosmetics differently. Additionally, what does it mean to “treat” something as food? I read it to refer to things like manufacturing, labeling, and storing Hemp-CBD, but I could be wrong. Licensing and Seeds The WSDA would issue hemp producer licenses. Current hemp licensees could transfer into Washington’s new program once it’s up and running. Hemp producers will only be allowed to use seeds listed in SB 5719 or approved by the WSDA. The bill also contains a clever provision that distinguishes seed varieties based on THC percentage. Certain seed “cultivar” (i.e., “a variation of the plant Cannabis sativa L. that has been developed through cultivation by selective breeding”) will be exempt from THC testing. WSDA will have the authority to approve seed cultivar and determine whether THC testing is required. Cultivar that were brought into Washington state before January 1, 2022 so long as “the state has planting, growth, and stability records covering at least three years.” Even though seeds will be regulated fairly robustly, SB 5719 does not indicate the need for seed-to-sale traceability as the WSDA will not be responsible for determining whether a hemp product was derived from an approved hemp cultivar. Regulatory Authority Though SB 5719 is a major overhaul, WSDA would still have authority over hemp in the Evergreen State. Both marijuana and hemp are ways to describe cannabis so it’s natural to wonder what is the LCB’s role in regulating hemp under SB 5719? SB 5719 specifically states that “all rules relating to hemp, including any testing of hemp, are outside the control of the [LCB].” However, the WSDA is mandated to consult with the LCB to establish rules and policies to prevent cross pollination between marijuana and hemp crops. In the event that a documented cross-pollination erupts between two farms growing hemp or marijuana, the farm operating first will be the victor. This first-in-time law would give marijuana farmers a massive upper-hand, at least initially, as Washington’s marijuana program is older and much larger than the hemp program. The WSDA and state lawmakers will undertake a task force for determining the need for crop insurance. Bottom line SB 5719, if passed in its current form, would drastically change Washington’s hemp laws. Like the 2018 Farm Bill, it’s a much more commercial friendly scheme. If you agree or disagree with the current version of SB 5719, you can submit comments here. If SB 5719 becomes law, it will take effect immediately. Given that there are several time-sensitive provisions in the bill, it makes sense to start planning now. We’ll keep an eye on this and other hemp-bills in Washington and other states. Source: https://www.cannalawblog.com/washington-cannabis-the-proposed-hemp-overhaul-is-here/ Washington Cannabis: The Proposed Hemp Overhaul is Here was originally seen on Felicia's Smokin' Blog from https://www.feliciasullivan.com/2019/02/07/washington-cannabis-the-proposed-hemp-overhaul-is-here/ |
ABOUT USAround Here, It's ALWAYS 4/20! ArchivesNo Archives Categories |






 In light of the recent federal
In light of the recent federal 
 RSS Feed
RSS Feed
